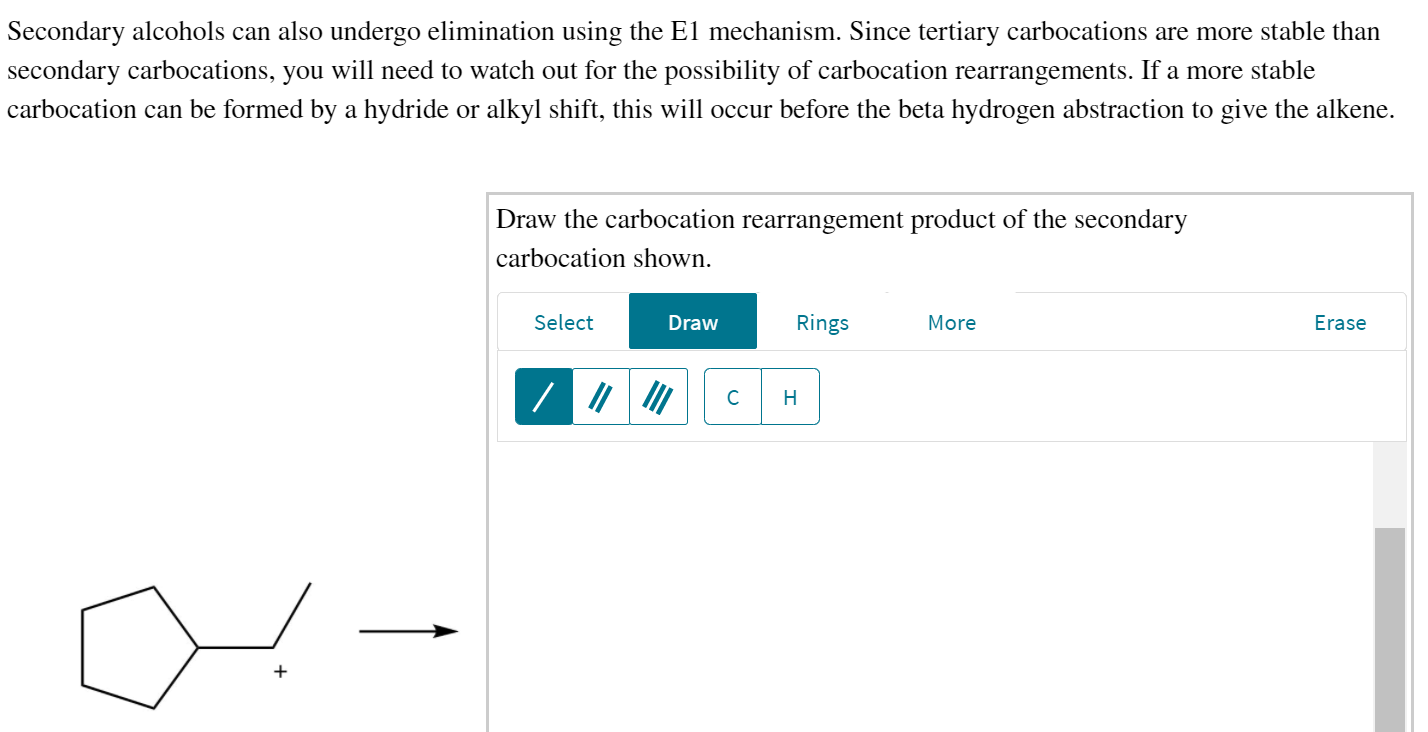
Draw the Carbocation Rearrangement Product of the Secondary Carbocation Shown.
E1 elimination reactions which will be covered in the next section can also include a hydride or alkyl shift leading to a more substitued alkene. Carbocation rearrangements can be defined as the movement of the carbocation from an unstable state to a more stable state by making use of different structural reorganizational shifts within the molecule.

E1 Energy Diagram Transition State Forming A Double Bond Reactions Energy Activities Chemistry
The confusion may be due in part to the continued use of the terms edge-protonated and corner-protonated to describe PCP.
. Step-by-step explanation It is known that alcohol undergoes dehydration if it is heated in the presence of acid. In the above example you can see that we get a primary carbocation at first. If a rearrangement is possible you should rearrange the molecule in order to obtain the major product.
When treated with acid mathrmC-mathrmOH bond is cleaved heterolytically to form a carbocation which. Draw the carbocation rearrangement product of the secondary carbocation shown. So this is a secondary carbocation and we think about the possibility of a rearrangement.
They are the most common carbocation. Next we think about the possibility of a rearrangement. Draw the carbocation rearrangement product of the secondary carbocation shown.
So this is the rearrangement that we would see. And we know from the previous video that a tertiary carbocation is more stable than a secondary carbocation. When are rearrangements possible.
Were going from a secondary carbocation to a tertiary carbocation via a hydride shift. And let me draw in the plus one formal charge on the carbon in red. Now if you look at one of the carbon atoms next-door a hydrogen can migrate with its pair.
So on this carbon in magenta theres still a hydrogen and a hydride shift would give us a. If a more stable carbocation can be formed by a hydride or alkyl shift this will occur before the beta hydrogen abstraction to give the alkene. Unlike secondary and tertiary alcohols the dehydration reaction occurs under an E2 elimination instead of an El mechanism due to the instability of the primary carbocation that would be formed.
However if a hydrogen moves from the secondary carbon to the primary we can get a secondary carbocation. So in the carbocation and rearrangements video another shift that we did was a hydride shift. Draw the carbocation rearrangement product of the.
But the goal is to form a more stable carbocation in a rearrangement. Finally lets do one more kind of carbocation rearrangement. Modeling of product distributions in biomasspetroleum refining5 This paper clarifies the role and structures of PCP in carbocation rearrangement supported by a large body of new computational results.
However alcohol B gives a less stable 2 carbocation at least initially. Most are migrations between adjacent atoms and are called 12-shiftsCarbocation rearrangements occur more frequently on secondary carbocations to form tertiary which are more stable and energetically more favorableIn general the bonding electrons of a. Draw the carbocation rearrangement product of the secondary carbocation shown.
If a secondary carbocation is vicinal to a tertiary carbon bearing a hydrogen a 12-hydride shift should occur. So this carbocation is secondary. This ones called a methyl shift.
And we go from a secondary carbocation on the left to a tertiary carbocation on the right which we know is more stable. In a rearrangement a group moves from one atom to another in the same molecule. So a less stable carbocation eg 1 or 2 might undergo a rearrangement reaction to form a more stable carbocation 2 or 3.
As mentioned above any reaction involving a carbocation intermediate may be subject to rearrangement. And just like the previous examples we dont need to draw in the hydrogens. We could just show our.
If a secondary carbocation is vicinal to a quaternary carbon a 12-alkyl shift should occur. Alkyl carbocation is a carbocation comprising an alkyl group. Professor Davis shows several examples of how secondary carbocations can rearrange themselves by hydride shifts alkyl shifts and ring expansions to form mor.
Carbocation rearrangements in other types of reaction. Rearrangements reactions usually occur to increase the stability of a carbocation. Primary alcohols can also dehydrate to alkenes.
The rearrangement of the secondary carbocation to more stable tertiary carbocation is shown in explanation part. The carbon in red is this carbon and thats a secondary carbocation because the carbon in red is directly bonded to two other carbons which I just marked there in magenta. Preparation and reactions of alcohols Alcohols can by prepared from alkyl halides epoxides alkenes etc.
Select Draw Rings More Erase. The most common rearrangement reactions are 12-hydride shifts and 12-alkyl shifts. The rate of this step and therefore the rate of the overall substitution reaction depends on the activation energy for the process in which the bond between the carbon and the leaving group breaks and a carbocation.
The different types of carbocation rearrangement are. Stability of carbocation intermediates. A hydrogen can move from one atom to the atom next door if it will increase the stability of the carbocation 3 0 2 0 1 0.
Since tertiary carbocations are more stable than secondary carbocations you will need to watch out for the possibility of carbocation rearrangements. We know that the rate-limiting step of an S N 1 reaction is the first step formation of the this carbocation intermediate. Alcohol A gives a reasonably stable 3 carbocation which reacts with the electron-rich benzene ring in a FriedelCrafts alkylation to give the product.
For instance rearrangement will be highly favoured if there is a conversion of a secondary carbocation can be formed from a primary carbocation The reason is simple because the carbocation is more stabilized in secondary carbocation than in a primary carbocation. Now I want to talk about one of the most interesting but also annoying things that carbocations do and thats called the carbocation rearrangement. This is known as a hydride-shift.
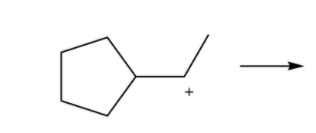
So this is a secondary carbocation. It turns out that carbocations are going to be able to rearrange to more stable positions if theyre adjacent to the carbocation and if it has more R groups than the carbocation has at the moment.

Solved Secondary Alcohols Can Also Undergo Elimination Using Chegg Com
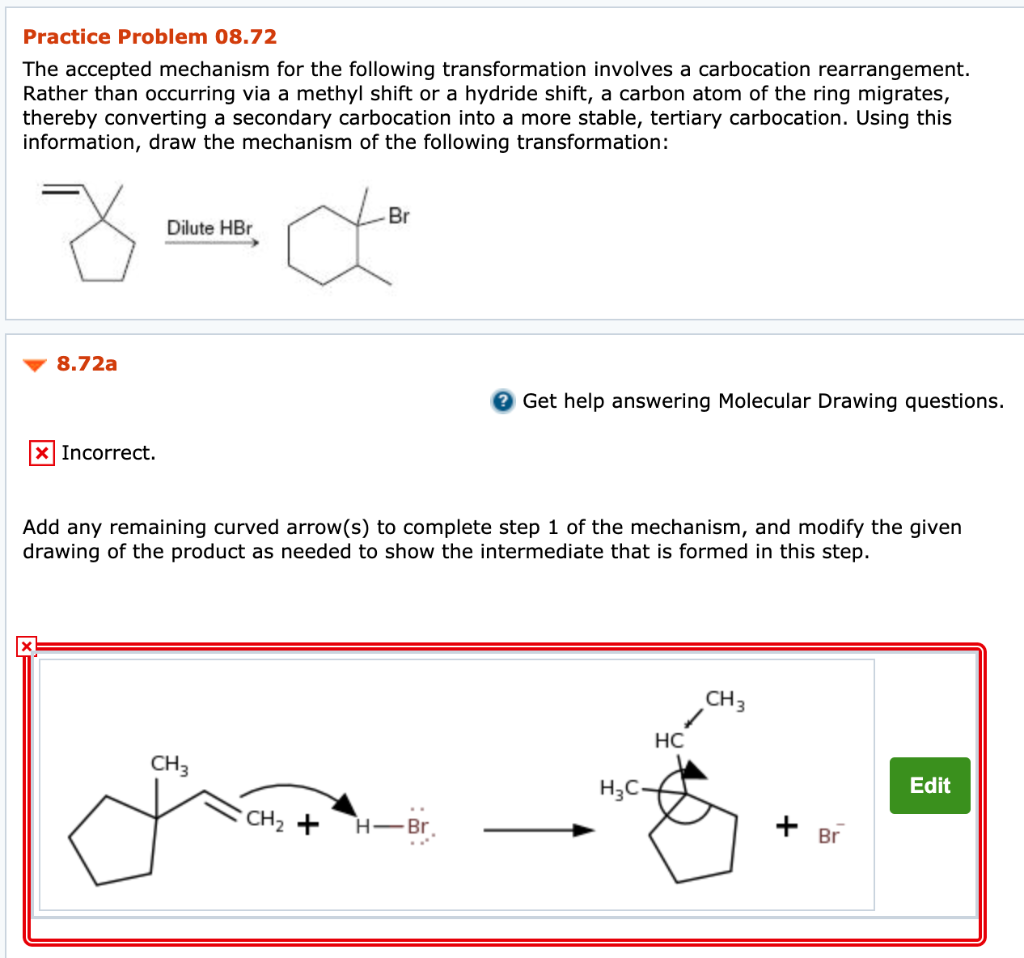
Solved Practice Problem 08 72 The Accepted Mechanism For The Chegg Com
Comments
Post a Comment